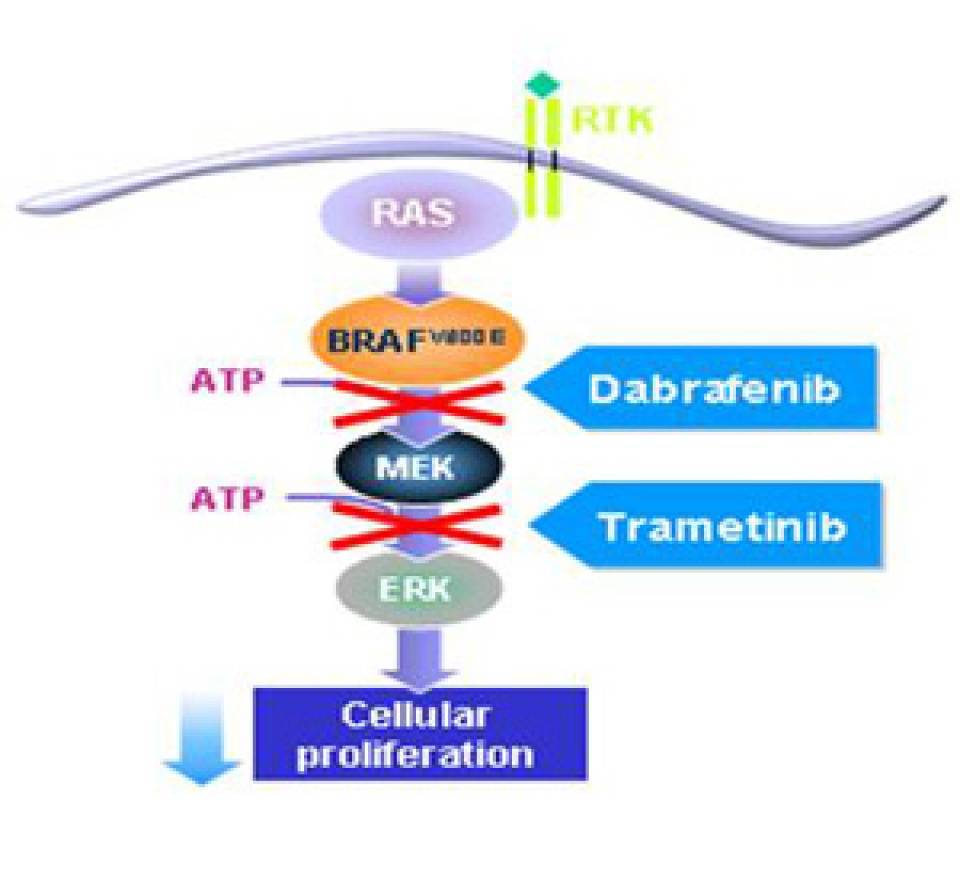
Metastatic melanoma is associated with a poor prognosis with a median overall survival of less than a year. Patients with this type of melanoma had no treatment options until the appearance of BRAF inhibitors three years ago. These inhibitors block the MAPK pathway and improve the overall survival of patients with BRAF V600 mutation compared to those receiving conventional chemotherapy. However, most patients develop resistance to treatment and the MAPK pathway is reactivated. Independently, MEK inhibitors administered in monotherapy have also proven to be effective in such patients.
This study compares the efficacy of the combination dabrafenib and trametinib (BRAF and MEK inhibitors, respectively) versus dabrafenib alone, as in pre-clinical trials has shown to delay the onset of resistance to treatment.
The study involved 423 previously untreated patients who had a metastatic melanoma with one of the most prevalent BRAF mutations: the V600E or V600K. They were randomly assigned to receive either the combination or the single agent therapy. The results showed that the combination of the two inhibitors results in a 25% relative reduction in the risk of disease progression and improves the survival in such patients. Dr. Ana Arance says: "These results are attributed to the inhibition of the MAPK pathway at two different nodes with the combination therapy, which delays the emergence of resistance to treatment and results in a significant clinical benefit for patients with advanced melanoma BRAF-mutated". Thus, the study published in the NEJM opens the door for the treatment of a type of tumor for which limited therapeutic options are currently available.
Hospital Clinic, the first reference center designated by CatSalut to attend cases of metastatic melanoma, was the first in having this type of medication. Dr. Aleix Prat, Head of the Medical Oncology Department, adds: "In the field of metastatic melanoma, the Clinic is the most experienced Spanish hospital and the Medical Oncology Department is the only one that has participated in all the clinical trials which led to the registration of new drugs to treat this disease".
Article reference:
Combined BRAF and MEK Inhibition versus BRAF Inhibition Alone in Melanoma
Long GV, Stroyakovskiy D, Gogas H, Levchenko E, de Braud F, Larkin J, Garbe C, Jouary T, Hauschild A, Grob JJ, Sileni VC, Lebbe C, Mandalà M, Millward M, Arance A, Bondarenko I, Haanen JB, Hansson J, Utikal J, Ferraresi V, Kovalenko N, Mohr P, Probachai V, Schadendorf D, Nathan P, Robert C, Ribas A, DeMarini DJ, Irani JG, Casey M, Ouellet D, Martin AM, Le N, Patel K, Flaherty K.
N Engl J Med. 2014 Sep 29. [Epub ahead of print]