The study, led by Balbino Alarcón from the Centre for Molecular Biology "Severo Ochoa" (CBMSO), has been published in Science Translational Medicine and has involved the participation of 32 Spanish researchers from CSIC, IDIBAPS, the Spanish National Center for Cardiovascular Research (CNIC) and Hospital de la Princesa in Madrid, mainly. In the case of IDIBAPS, the team of Pablo Villoslada, head of the group Multiple Sclerosis, Pathogenesis and New Therapies of the Neuroimmunology program of IDIBAPS, has participated. They have performed the trials in animal models for multiple sclerosis.
The new experimental drug is an inhibitor of the activation of T lymphocytes, the ones in charge of detecting the antigens (target proteins) of the organism, both the present in organs and tissues and the strangers (those that pose a threat and a potential infection). For their work, T lymphocytes have an essential receptor: the T cell receptor (TCR). The TCR transmits information inside the lymphocyte to indicate that it has detected or recognized an antigen (external or not), an information that is transmitted through different proteins that are inside the cell. One of them is the NCK. When TCR recruits NCK or binds to it within the cell it transmits information indicating that it has recognized an antigen. The drug inhibits or blocks the interaction between TCR and NCK and, as a result, makes T lymphocytes no longer respond to their own antigens in an autoimmune disease but does combat the external antigens of an infectious process. It is their differential effect, explain the researchers leading the study.
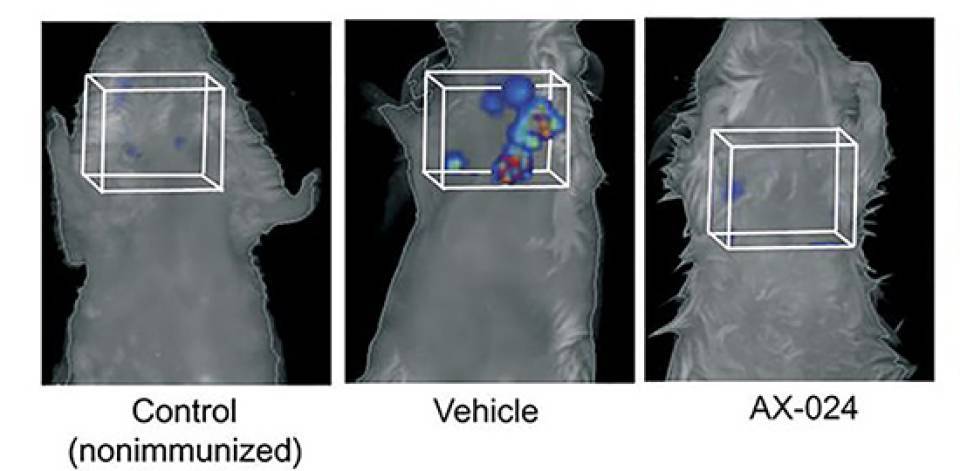
The new drug has been tested on healthy volunteers and has passed the toxicity tests. In mouse models, moreover, it has been found to have effects even when the compound is discontinued, which means that a treatment for a limited time may be enough for the remission of the disease’s symptoms, avoiding the need of a lifetime medication.
Article reference:
First-in-class inhibitor of the T cell receptor for the treatment of autoimmune diseases
Aldo Borroto, Diana Reyes-Garau, M. Angeles Jiménez, Esther Carrasco, Beatriz Moreno, Sara Martínez-Pasamar, José R. Cortés, Almudena Perona, David Abia1, Soledad Blanco, Manuel Fuentes, Irene Arellano, Juan Lobo, Haleh Heidarieh, Javier Rueda, Pilar Esteve, Danay Cibrián, Ana Martinez-Riaño, Pilar Mendoza, Cristina Prieto, Enrique Calleja, Clara L. Oeste1 Alberto Orfao, Manuel Fresno, Francisco Sánchez-Madrid, Antonio Alcamí, Paola Bovolenta, Pilar Martín, Pablo Villoslada, Antonio Morreale, Angel Messeguer and Balbino Alarcon.