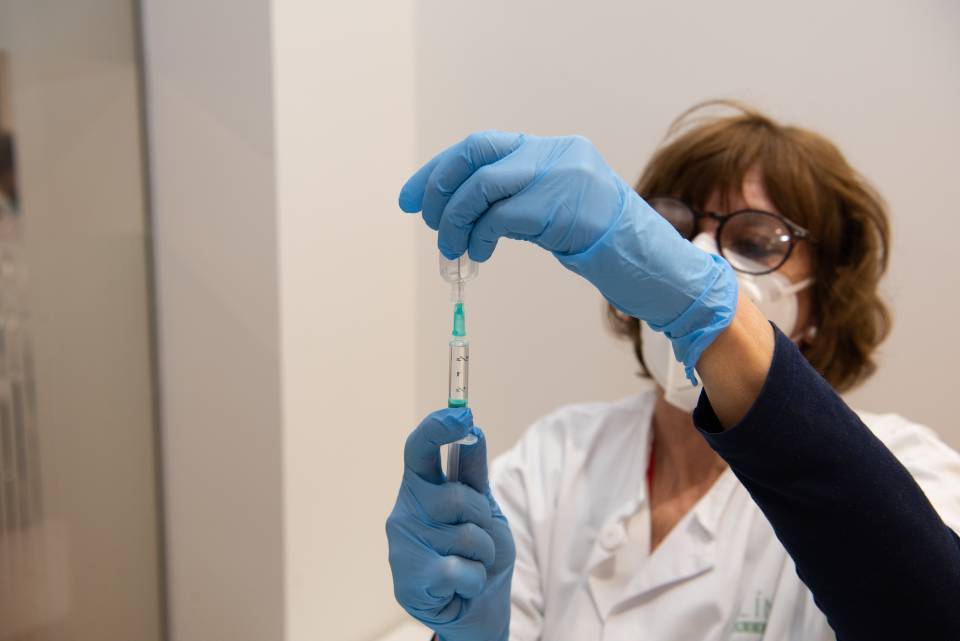
Volunteers will receive two doses of the new investigational vaccine or of a vaccine that is already on the market. They will be monitored for up to one year in order to study the vaccine’s safety and response (immunity).
The trial is being assessed by the Ethics Committee of the Hospital Clínic de Barcelona and the Spanish Agency of Medicines and Medical Devices (AEMPS) and will start in August 2021 at the Hospital Clínic de Barcelona and the Hospital Universitari de Girona Dr. Josep Trueta.
The volunteers will be paid.
If you are interested, please contact:
Hospital Clínic de Barcelona
Telephone: 634 264 791
e-mail: cvac@clinic.cat
Hospital Universitari de Girona Dr. Josep Trueta
Telephone: 638 686 116
e-mail: usrgirona@idiapjgol.info