Fats are essential for life, but very toxic in excess. All the cells contain lipid droplets (LDs), organelles that store the fatty acids excess allowing the cell to reduce its toxicity and supplying them when there is an energy requirement. Although its function is well described in adipocytes, the body’s cell subtype specialized in the accumulation of fat, little is known about the role of LDs in the other cells and the mechanism of interaction with other cellular organelles. The knowledge of these mechanisms would be very important since an excessive accumulation of LDs is the cause of diseases associated with obesity, such as type II diabetes, and atherosclerosis, metabolic syndrome and fatty liver disease.
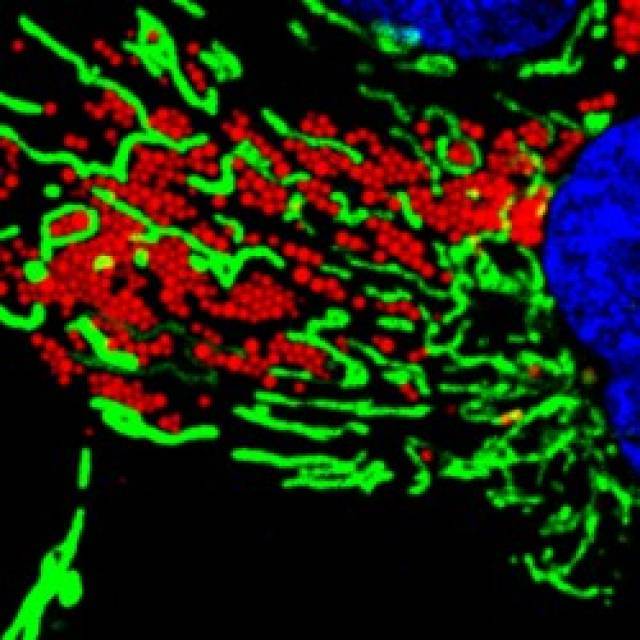
For all this to happen, the activation of an enzyme called AMPK (AMP-activated protein kinase), which is a cellular energy sensor that helps in the energy balance and calories consumption, is needed. Researchers have shown that AMPK acts simultaneously increasing the mobility of LDs, reorganizing the network of microtubules and activating mitochondria both in normal and starvation conditions.
Many diseases arise from a deficiency in the bioenergetic mechanisms of cells and LDs accumulation in different tissues is associated with several highly prevalent pathologies. This article shows which is the mechanism to mobilize LDs for energy production, both in normal and scarcity conditions. This discovery may deepen the understanding of the causes and treatment of diseases such as diabetes and atherosclerosis: "We have seen that with the administration of a drug such as metformin, one of the most used in the treatment of Type II Diabetes, all processes to mobilize LDs are triggered, allowing fat burning and bending energy production efficiency", explains Dr. Albert Pol, the study coordinator.
Article reference:
Albert Herms, Marta Bosch, Babu J.N. Reddy, Nicole L. Schieber, Alba Fajardo, Celia Rupérez, Andrea Fernández-Vidal, Charles Ferguson, Carles Rentero, Francesc Tebar, Carlos Enrich, Robert G. Parton, Steven P. Gross, Albert Pol
Nature Communications 6, Article number: 7176 doi:10.1038/ncomms8176

