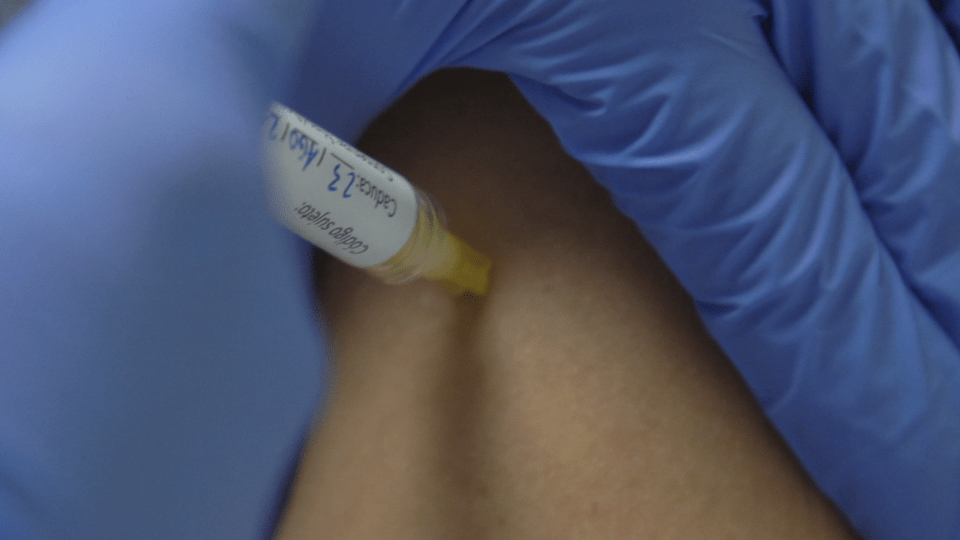
The first people to take part in Phase I/IIa of the HIPRA COVID-19 vaccine clinical trial received the first dose this week. Several participants have been inoculated this week, some of whom have received the study vaccine and others a commercially available vaccine. The rest will receive the vaccine in the next few days at the Hospital Universitari Clínic de Barcelona and the Hospital Universitari de Girona Doctor Josep Trueta. The trial was authorized by the Spanish Agency for Medicines and Health Products (Agencia Española de Medicamentos y Productos Sanitarios [AEMPS]) and by the Ethics Committee of the Hospital Universitari Clínic de Barcelona.
In this phase, the safety of the HIPRA COVID-19 vaccine is being evaluated, as is its immunogenicity. Some 500 people aged 18-39 responded to the call made in mid-July. Last week, the teams involved in the clinical trial selected 30 participants. This process involved a questionnaire and a blood test to ensure, amongst other things, that they had not been infected with COVID-19 and been asymptomatic and thus unaware of having the virus. The selected volunteers, both men and women, are healthy people who have not been infected with SARS-CoV-2 nor received any dose of a COVID-19 vaccine.
To carry out the trial, the participants were divided randomly into cohorts, which will receive different doses of the vaccine. The first cohort receives the lowest dose, and the doses are gradually increased in the following groups. Some of the volunteers will be randomly assigned to receive the HIPRA vaccine and the rest will receive a vaccine that is already commercially available. Neither the volunteers nor the research team will know which vaccine has been administered in each case. An independent data monitoring committee oversees the entire trial. One of the aims of the clinical trial is to determine the optimal dose of the vaccine, which is why it is carried out with a small group of people. After 21 days, the participants will receive the second dose.
Monitoring of the participants for one year
This phase is being carried out in a totally controlled manner. Throughout the trial, all participants are closely monitored, which will allow preliminary analyses to be carried out in order to be able to continue advancing with the research. When the clinical trial ends, all the data will be analysed and the final conclusions will be drawn. This monitoring, which lasts for one year, is performed by researchers from the two health centres through post-vaccination visits and follow-up visits.
If Phase I/IIa produces the expected results, it is hoped to advance to Phase IIb/III, which will have to be approved by the AEMPS.
The two hospitals and research teams would like to thank the public for the great response received to the call for volunteers, and would like to highlight the value of the contribution made by these individuals in helping science to advance.
HIPRA COVID-19 vaccine
The HIPRA COVID-19 vaccine is a recombinant protein vaccine designed to optimize its safety and generate a potent immune response that neutralizes the COVID-19 virus. It will be stored at between +2 and +8ºC.
The adjuvant used is an oil-in-water emulsion like the one that has been used for over 20 years in thousands of doses of influenza vaccines and has proven to be safe.
Research teams
The clinical trial for the HIPRA vaccine is being conducted by two research teams, with three principal investigators.
On the one hand, the Vascular Health Epidemiology Group from the Catalan Health Institute (Institut Català de la Salut) in Girona, led by Dr. Rafel Ramos, researcher at the “Jordi Gol” University Institute for Primary Care Research (Institut Universitari d'Investigació en Atenció Primària [IDIAP] “Jordi Gol”) and at the “Dr. Josep Trueta” Biomedical Research Institute of Girona (Institut d'Investigació Biomèdica de Girona Dr. Josep Trueta [IDIBGI]).
On the other hand, at Hospital Clinic of Barcelona is working a multidisciplinary team built up by medical, nursing and administration professionals of the Infectious Diseases and Preventive Medicine and Epidemiology Service. The professionals that lead the study are Dr. Àlex Soriano, head of Infectious Diseases Service and Head of the nosocomial infections group at the Biomedical Research Institute August Pi i Sunyer (IDIBAPS), and Dra. Lorna Leal, specialist in the infectious diseases service and member of the AIDS and HIV Infection group of IDIBAPS. It must also be underlined Clara Castán task. At the same time, it’s to be highlighted all the work that has been done and is being done from the Clinical Trials Unit (CTU) of the Clinical Farmacology Service (it’s important to point up the professionals’ work of Judit Pich and Joan Albert Arnaiz at CTU that have been collaborating in this project). CTU’s main goal is to provide support on the ethical, methodological, regulatory, and logistical aspects of clinical trials with drugs or advanced therapies promoted by researchers at the institution.
Another institution involved in the trial is IrsiCaixa, where various research teams are participating in the clinical trial carrying out a significant part of the assessment of the immune response to the study vaccine, along with IDIBAPS.