The mitochondria are organelles found in the cells and carry out different functions, the best-known of which is generating chemical energy for the cell; however, they also play a role in the cell cycle and cell death. Malfunction of the mitochondria is linked to different diseases.
“One of the objectives of our group is to understand the mitochondrial mechanisms involved in neuronal regulation of hunger and body weight”, explained Marc Claret, head of the IDIBAPS research group Neuronal control of metabolism. “Earlier studies published by us have shown that abnormalities in the mitochondria of a specific group of neurons favors the onset of diabetes and obesity”.
In a new study, published in the journal Cell Metabolism and highlighted on its cover page, the researchers focused on understanding the role of the mitochondrial cristae, the internal folds in the internal mitochondrial membrane, in a population of neurons in the hypothalamus, called POMC, which are essential for systemic regulation of the energy balance.

To do this, they carried out different experiments: “We used electron microscopy to analyze the shape of the mitochondrial cristae in POMC neurons at different energy states and we generated genetically modified mice with mitochondrial cristae that did not produce the OPA1 protein, which is key to the maintenance and dynamics of the mitochondrial cristae. “We studied both the physiology and the molecular mechanisms of lipolysis (i.e., fat mobilization) of adipose tissue and the beneficial effects that regulating the entry of calcium into the mitochondrion has on the phenotype of these mice”, explained Alicia Garcia, the lead author of the study.
The results indicate that the energy status of the organism, i.e., whether it is fasting or has been fed, whether it has had a normal diet or a fat-rich diet, modulates the shape of the mitochondrial cristae in POMC neurons. Moreover, eliminating the OPA1 protein causes obesity in mice. This obesity is caused by a defect in the activation of lipolysis in the adipose tissue during fasting.
“What we have seen is that alterations to lipolysis and body weight occur due to deregulation of the entry of calcium into the mitochondria of the POMC neurons, which leads to an overload of this ion, preventing the correct functioning of the mitochondrion and of the neuron”, added Alicia García. Furthermore, “pharmacologically blocking the entry of calcium into the mitochondrion normalizes the observed abnormalities in lipolysis and body weight”, concluded Claret.
This study was carried out with the support of the European Research Council and the BBVA Foundation.
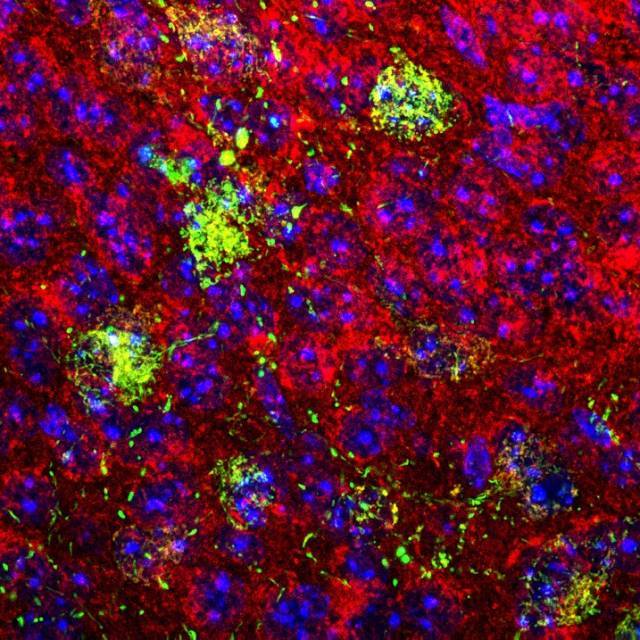
Confocal microscopy image of a coronal section of mouse brain showing the mitochondria in red and the mtGCamP6s mitochondrial calcium sensor expressed in POMC neurons
Article reference:
Alicia G Gómez-Valadés, Macarena Pozo, Luis Varela, Mehdi Boutagouga Boudjadja, Sara Ramírez, Iñigo Chivite, Elena Eyre, Roberta Haddad-Tóvolli, Arnaud Obri, Maria Milà-Guasch, Jordi Altirriba, Marc Schneeberger, Mónica Imbernón, Angela R Garcia-Rendueles, Pau Gama-Perez, Jonathan Rojo-Ruiz, Bence Rácz, Maria Teresa Alonso, Ramon Gomis, Antonio Zorzano, Giuseppe D'Agostino, Clara V Alvarez, Rubén Nogueiras, Pablo M Garcia-Roves, Tamas L Horvath, Marc Claret
DOI: 10.1016/j.cmet.2021.07.008